Primary Literature Figures – Chemistry
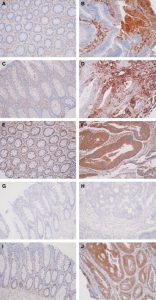
In chemistry class, we’re not only learned formulas and labs. We also did some research that university students are learning. We looked at the figure about normal, colorectal tumor, and colorectal cancer cells analyzed for intensity of annexin protein staining. Also, we looked at the picture figure about panels A, C, E, G, and I (left) are normal tissue, Panels B, D, F, H, and J (right) are colorectal cancer tissues. A and B show annexin A1, C and D show annexin A2, E and F show annexin A4, G and H = annexin A7, and I and J = annexin A11.
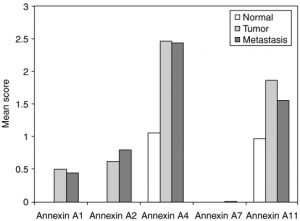
After all of the in-depth look at the two figures in group, we try to interpret it. One example we try to figure out the title of the figures. Our hypothesis for the title is Cancer Tissues Appear to Have More Annexin (A1, A2, A4, A11) Compares to Normal Tissues. We discussed the question for this research is “between normal cells and cancer cells, which one has more annexin?”
What we learned from our background and research information is that MMP (Matrix metalloproteinase) is a group of enzymes that create a microenvironment for cancer cells to grow and break down ECM (Extracellular Matrix). In order for those tumor cells to grow, they need nutrient, so MMPs also help to increase blood vessels for the nutrient to flow through the blood vessel. These enzymes are able to control annexin, which are proteins that can be found in eukaryotic organisms; from that conclusion, there’s an evidence that it is possible for the annexin to grow in tumor cells. Our we conclude this is that tumor cells have more annexin than normal cells. The amount of annexin A4 and A11 in the tumor cells are almost twice as much as the normal cells. Annexin A4 has the most protein present in both types of cells. However, there is no Annexin A7 in both types of cells. We understand more about science literature especially interpret the figure by ourselves, and predict what are the scientists’ prediction or what are their results for the experiment.
Tumor Figure 1
Tumor Figure 2
First Flame Lab Test
When my facilitator asked, “Do you like chemistry?” I replied honestly “No. I don’t like it.” But my thought changed in a week after I learned chemistry. It’s not boring at all. One of the activities I love the most in chemistry was doing labs. One of the labs is called the “”Flame Test Lab”.
This is the introduction of the lab.
Flame tests provide a way to qualitatively test for the presence of specific elements by seeing colored flames. The color we see is a combination of the visible wavelengths of light emitted by the atoms. Each element has a different “pattern” of electrons so it will show a different combination of colors.
In this lab performed flame tests on seven different elements. You tried to observed to identify an unknown solution.
I have so much fun seeing the flame color that changes because of different chloride solutions. My group, we took a turn to test the flame, and while we did our experiment we write down results and observation. Based on our experiment we found out the unknown chloride is Lithium.